
“Weighing” In On Hormones
Everything we read these days warns us that Americans are overweight. According to statistics published by the National Institutes of Health, roughly 55 percent—more than half of the United States’ population—is overweight, or even obese. This problem is considered an epidemic; it is our number one disease.
In today’s culture, which touts countless diet fads, diet books, and new exercise regimens, the issue of weight control is everywhere. In Dr. Scott Isaacs’ book, Hormonal Balance, he points out that, in so many cases, the basis for dieting falls short because it often does not include hormones as a component of weight issues. Hormones indeed play a very critical role in weight control. In addition, our individual genetics, lifestyle, and exercise regimens factor into the battle to keep our metabolism going strong.
There is no doubt that our hormone production and metabolism slow down as we age. That is why it makes sense to consider how hormone levels may be affecting changes in your own weight and metabolism. A healthy and proper hormonal balance is helpful in getting a handle on weight gain, especially during phases of life when our hormone levels may fluctuate. As more research is carried out on the way in which specific hormones impact our metabolism, it becomes clear that the balance of hormones may be a key to gaining the upper hand in the battle of the bulge.
In order to understand how various hormones in our body influence our weight, let’s begin by taking a look at what some of these hormones actually are, wherein the body each exerts its effects, and what each hormone actually does in relation to weight and metabolism.
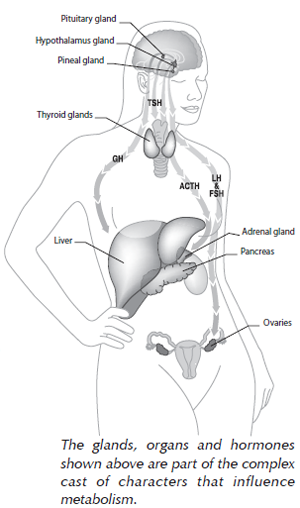 Pituitary Gland-Controlled Hormones
Pituitary Gland-Controlled Hormones
Our bodies are hormone-producing machines. And it is not only the major glands but virtually every organ in our body that makes hormones. Most of the hormones we’re familiar with are controlled by a very important “master gland” located in our brain: the pituitary gland. The pituitary gland itself is controlled by a part of the brain called the hypothalamus.
As a master gland, the pituitary gland produces hormones that regulate several other glands in the body including thyroid-stimulating hormone (TSH), which stimulates the thyroid, and adrenocorticotrophic hormone (ACTH), which regulates adrenal gland hormone production, including cortisol and dehydroepiandrosterone (DHEA). The pituitary gland also controls the production of hormones such as luteinizing hormone (LH) and follicle-stimulating hormone (FSH) which, in turn, influence hormones made by the reproductive glands (i.e., progesterone, testosterone, and estrogens). The pituitary gland’s secretion of growth hormone regulates hormones made in the liver.
Hormones secreted by all the glands in this somewhat complex web communicate by sending signals back to the master gland in a fine-tuned system called “feedback.” As we’ll discuss in the following sections, a breakdown in the communication between any of these hormones can affect your body’s metabolic rate and weight.
Other Major Hormone-Producing Glands
Our bodies contain other hormone-producing glands that are not controlled by the pituitary gland. The pancreas makes the hormones insulin (more discussion below), glucagon, and somatostatin, which are all tied to the production, storage, and metabolism of sugar in our bodies. The proper function of the pancreas is therefore very important in the battle to maintain a healthy weight.
Other hormones in the body not regulated by the pituitary gland include cholecystokinin, which is made in the intestine and signals when the stomach is full, and a more recently discovered hormone called leptin, which is produced by fat cells. Finally, the brain makes many hormones including serotonin, melanocyte-stimulating hormone (MSH), and melatonin.
How Hormone Changes and Imbalances May Affect Weight
With so many glands producing so many hormones—many of them intricately connected in a communication system that is necessary to keep our bodies functioning at a healthy level— it is not surprising that a slight alteration can throw everything out of whack. Often, one of the first signs of this is a change in metabolism, which may lead to a change in weight.
Insulin and Hormones of the Pancreas
When we think of weight gain, we usually associate it with excess food or lack of exercise (or both). The hormone most closely linked to food is insulin, and insulin is a critical player in the way our body uses the fuel provided by food and its components, i.e., carbohydrates, proteins, and fats.
The pancreas is the organ that makes insulin, as well as the hormones glucagon and somatostatin. These hormones are secreted into the blood and act throughout the entire body. Glucagon is a digestive hormone that is naturally released from the pancreas when food, particularly protein, leaves the stomach and enters the small intestine. Glucagon has several jobs to do including slowing the exit of food from the stomach, which makes you feel full. It also acts to raise blood sugar and signals the liver to excrete glucose or sugar. An imbalance in glucagon levels can thus wreak havoc on the metabolism of food and influence weight gain or loss.
Somatostatin has an important regulatory role as it can block the secretion of insulin and glucagon. In fact, its main function in the pancreas is to shut down the specialized cells responsible for insulin secretion.
Other hormones made by the pancreas differ in that they are secreted directly into our intestines and aid in the process of digestion. All of these hormones work together to help control our metabolism; it is not surprising that an imbalance of any of these hormones, especially insulin, might affect our body weight.
Because insulin works by increasing our appetite and promoting the storage of nutrients such as fat, insulin can promote weight gain. When insulin levels are high, it can be very difficult to prevent weight gain. Insulin resistance is a common hormonal imbalance in people who are overweight and cause high insulin levels because the receptors for insulin (which are proteins on the surface of cells that respond to the actions of insulin) do not work efficiently. This results in malfunctioning of insulin’s normal action; people with insulin resistance require higher insulin levels to maintain their metabolism. A rise in insulin levels is then often accompanied by a gain in weight.
A type of insulin resistance that is gaining more attention lately has been labeled Metabolic Syndrome or “Syndrome X.” This syndrome is characterized by a variety of dangerous health conditions including diabetes, obesity, high blood pressure, high cholesterol, and heart disease. Because an imbalance in the hormone insulin facilitates these syndromes, it is important to check insulin levels if any of these symptoms develop.
Many hormones are involved in the body’s metabolism and can affect weight control, including those listed below:
| Gland/Organ: | Hormones Produced: |
| Pituitary | TSH (stimulates thyroid)
ACTH (regulates adrenal gland) Growth Hormone (regulates liver hormones) LH and FSH (regulate reproductive gland hormones) |
| Thyroid | Thyroid hormones T3 and T4 |
| Adrenal | Cortisol
DHEA |
| Pancreas | Insulin
Glucagon Somatostatin |
| Ovaries/Testes (Reproductive Glands) | Estrogens
Progesterone Testosterone |
| Intestine | Cholecystokinin |
| Fat (adipose) tissue | Leptin |
| Brain | Serotonin
MSH Melatonin |
Testosterone
The onset of andropause in men is associated with a decrease in testosterone levels. Testosterone helps maintain strong and healthy bones and muscles, and low testosterone has been linked to increased amounts of fat in the body. According to Dr. Malcolm Carruthers in The Testosterone Revolution, “weight gain which doesn’t respond to diet is one of the most common and demoralizing effects of this condition.”
Dr. Carruthers explains that a spreading waistline may be attributed in part to the body’s increased resistance to the action of testosterone and because more testosterone is converted to estrogen. Testosterone deficiency is also responsible for more sugar and protein from food to be converted to fat, so the natural tendency is to gain weight. Testosterone therapy may, therefore, help to improve sugar metabolism by directing more to the rebuilding of muscles, rather than to the conversion to fat.
On the other hand, a hormonal condition called polycystic ovary syndrome (PCOS) that affects millions of women results in an overabundance of certain hormones including testosterone. One of the most common symptoms of PCOS is weight gain, so in these cases, an excess of testosterone may be a culprit.
Estrogens
One of the most common complaints voiced by women as they experience menopause is that they gain unwanted weight. While this is certainly not the case for everyone— and it can happen before, during, or after menopause— weight gain is frequently associated with the changes in hormone levels that occur during menopause. Declining estrogen levels may be only one factor involved; others include heredity, diet, physical activity, and weight distribution.
However, fat distribution has been shown to change as a result of decreasing estrogens. In particular, as estradiol production declines and estrone levels rise during and after menopause, women tend to show excess fat in a pattern similar to that seen in men—it appears around the belly (often described as “apple-shaped”).
As a woman’s ovaries naturally produce less estrogen during perimenopause, fat cells in the body respond by producing more estrogen. This is the body’s way of striving to maintain hormonal balance, but it can result in an increase not only in the number of fat cells, but their size as well. Estrogen is capable of stimulating the enzymes in fat cells and directing where fat is stored. Often it is sent right to the waistline.
The interaction between estrogens and fat cells may help to explain why menopause and its associated drop in estrogen production are often a time of weight gain, or fat redistribution. While estrogen therapy does not necessarily help women lose weight, it may help shift fat back to the chest and hips, rather than the stomach.
Dr. Isaacs explains that “what estrogen can do is improve vitality and insulin resistance, two key factors in battling the bulge.” He also states that studies confirm that women who take estrogen have more muscle and less fat than those who do not.
Thyroid
The thyroid gland and the hormones it produces may have a wide-ranging impact on our weight because the thyroid controls, among many other things, our metabolism. When the thyroid begins to malfunction and thyroid hormone levels decline, energy levels typically decrease, causing a feeling of sluggishness. This also makes you less motivated to exercise. These effects, coupled with a lowered metabolism, often lead to weight gain.
Dr. Mark Starr, in his book Type 2 Hypothyroidism, believes that there is a strong connection between the obesity epidemic in the U.S. and the growing incidence of type 2 hypothyroidism. He defines this as low thyroid function due to an insensitivity of tissues to thyroid hormones, which appear as normal levels in the blood.
Compounded with a low metabolic rate creating weight problems, type 2 hypothyroidism often causes tissue accumulation of mucin (a gel-like substance normally found in the skin) and further contributes to the appearance of fat deposits.
Cortisol
Cortisol is a hormone that plays a role in the regulation of blood sugar levels and determines immune system response. A primary function of cortisol is to stimulate the conversion of protein to glucose; this is critical for maintaining appropriate levels of blood glucose for use throughout the body.
Fluctuating glucose levels may have a direct effect on signals sent to the brain regarding hunger and appetite. Cortisol production also stimulates the release of fatty acids in adipose (fat) tissue, but the long-term effect is often a gain in fat.
Cortisol is considered the body’s primary stress hormone as it provides responses to stresses of all types—and we all know how stress can influence appetite and eating. Chronic stress is known to cause sustained high levels of cortisol, and one of the many effects is an increase in appetite and cravings for certain foods. In his book The Cortisol Connection, Dr. Shawn Talbott says, “because one of the primary roles of cortisol is to encourage the body to refuel itself after responding to a stressor, an elevated cortisol level keeps your appetite ramped up—so that you feel hungry almost all the time.”
Melatonin
Another hormone that is believed to influence metabolism and weight control, although indirectly, is melatonin. This hormone is produced by the pineal gland, a small gland embedded deep in the brain that controls the body’s biological clock.
Around the age of 40, the production of melatonin by the pineal begins to slow down. Because melatonin has been shown to decrease cortisol levels in the body–and cortisol is produced in response to stress–melatonin may help regulate the sometimes harmful effects of cortisol.
With regard to metabolic control, melatonin has also been shown to enhance the production of T3, which is important in the regulation of healthy metabolism. That is why changes in melatonin levels can indirectly affect some of the body’s mechanisms that regulate metabolism, stress, and weight gain.
Human Growth Hormone
Recent studies have demonstrated that there is an inverse correlation between body fat and human growth hormone (HGH) production. In general, the more body fat you have, the less growth hormone you release; conversely, the less body fat you have, the more growth hormone you release. But why does this matter? According to Dr. Ronald Klatz, in his book Grow Young with HGH, for older people, trying to lose central fat from around the belly is very difficult. Adequate growth hormone may actually help in the battle against fat accumulation.
Studies also suggest that a decrease in growth hormone secretion in overweight individuals who carry their weight mostly around their belly is a sign that their hormonal system—especially the regulation of growth hormone and other related hormones— is not functioning properly. One such study showed that when this problem is treated by providing the body with growth hormone, other benefits such as better regulation of cholesterol levels, blood pressure, and glucose metabolism may occur.
Leptin
Leptin, a very recently discovered hormone, is proving to be a significant player in the overall system of hormones and their relation to weight, body shape, appetite, and metabolism. Secreted by fat cells contained in white adipose (fat) tissue, leptin has been shown to be a master regulator of many other hormones, including the sex hormones and those produced by the thyroid and adrenal glands.
Leptin works by sending a signal to our brain to let it know how much fat is stored, analogous to a fuel gauge in a car telling us how much gas is in the tank. The information that leptin sends to our brain allows the brain to act accordingly and regulate energy for our entire body. When the signaling by leptin malfunctions, weight control may be affected in either of two ways. One problem can occur when fat cells producing leptin don’t produce enough, so leptin isn’t able to signal to the brain that the body is satiated.
Another problem is a condition called leptin resistance and occurs in overweight people when leptin is no longer efficient at sending information to the brain. When the brain does not detect the proper level of leptin, it is fooled into thinking that fat storage levels are low and responds by slowing down the metabolic rate in order to store more fat as fuel. This results in excess fat in the body.
Leptin resistance is also believed to be responsible, at least in part, to the weight gain that many women experience after menopause. As estrogen levels begin to decline, it creates a breakdown in communication between estrogens and leptin, creating an imbalance in the signals involving leptin, estrogen, testosterone, and insulin.
Human Chorionic Gonadotrophin (hCG)
Back in 1954, endocrinologist Dr. Albert T. Simeons theorized that there are three types of fat in the body: structural fat such as the padding on the feet and the cushions between organs; fat reserves, which are easily accessed for energy when the body needs it; and abnormal fat deposits, which cannot be accessed and are a hallmark of obesity.
In his book Pounds and Inches, Dr. Simeons describes his experience with using small doses of hCG, in combination with a very low-calorie diet, to mobilize the abnormal fat deposits as a treatment for obesity. While the use of hCG for weight control is somewhat controversial, it is not surprising that the action of hCG on the hypothalamus, found in the most primitive part of the brain, may have something to do with how we store and use our fuel.
Hormone Balance and Your Weight
As discussed here, many different hormones play a role in weight control and metabolism. A shift in hormone levels can affect the delicate balance needed to maintain a healthy weight. In addition, obvious factors such as food intake, exercise, and lifestyle (e.g., stress, smoking, and alcohol consumption) also affect your weight, both directly and indirectly, because hormone production and function are also influenced by these other factors.
With all the complexities involved with the metabolism and weight control, you may want to partner with your healthcare practitioner to determine the best approach for achieving a weight that is right for you.
Connections is a publication of Women’s International Pharmacy, which is dedicated to the education and management of PMS, menopause, infertility, postpartum depression, and other hormone-related conditions and therapies.
This publication is distributed with the understanding that it does not constitute medical advice for individual problems. Although this material is intended to be accurate, proper medical advice should be sought from a competent healthcare professional.
Publisher: Constance Kindschi Hegerfeld, Executive VP – Women’s International Pharmacy
Co-Editors: Julie Johnson; Carol Petersen, RPh, CNP – Women’s International Pharmacy
Writer: Kathleen McCormick – McCormick Communications
Illustrator: Amelia Janes -Midwest Educational Graphics
Copyright © Women’s International Pharmacy. This newsletter may be printed and photocopied for educational purposes, provided that your copy (or copies) include full copyright and contact information.
 For more information, please visit womensinternational.com or call 800.279.5708.
For more information, please visit womensinternational.com or call 800.279.5708.
Women’s International Pharmacy | Madison, WI 53718 | Youngtown, AZ 85363

